Alpha-Stim
Treating Anxiety, Depression, and Insomnia with the Alpha-Stim® AID
The Alpha-Stim® AID is a Cranial Electrotherapy Stimulation (CES) device that uses low-level electrical current to safely and effectively treat anxiety, depression, and insomnia. Initially cleared by the FDA in 1992 as a prescriptive, noninvasive treatment, CES has an extensive safety record, with few side effects (less than 1%) and considerable scientific evidence of the significant results patients can achieve.
Alpha-Stim treatments are cumulative; however, most patients show at least some improvement after the first treatment. Alpha-Stim technology is safe, with no serious adverse events reported in over 30 years of clinical use.
The Foundation: A Patented Waveform
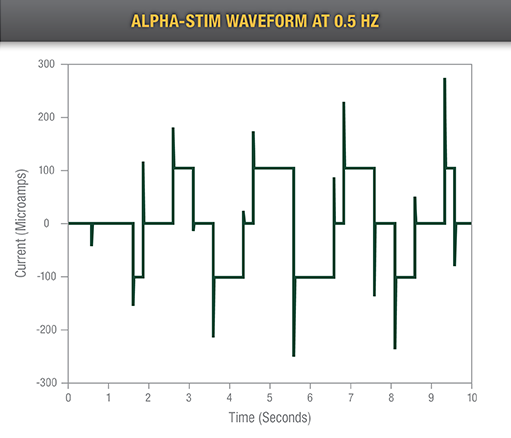
The only rigorously tested CES waveform designed and refined to improve effectiveness
Delivers a very low level of current in pulsed microcurrents of less than one milliampere
Effectively targets cell receptors, activating them through frequency matching in a manner similar to chemical ligands
The Impact: An Electrical Intervention
The brain functions electrochemically and can be readily modulated by electrical intervention1,2
Alpha-Stim impacts the electrochemical functions of the brain to improve patients’ emotional and physiologic states
Designed to effect the activity of subcortical brain structures known to regulate emotions
fMRI studies demonstrate the waveform current changes the appropriate brain structures
The Results:
Significant Improvement, Quickly, With Lasting Effect
Electroencephalographic studies show:
Increased alpha activity
Decreased delta activity
Decreased beta activity
As the leader in CES, Alpha-Stim has helped clinicians around the world offer fast, safe, and effective relief for patients suffering from anxiety, depression, and insomnia.
CLINICAL RESEARCH
Safety, Impressive Results, and Long-Term Effectiveness
Physician and patient surveys that show 90% of the people who use Alpha-Stim® get significant relief. This is supported by over 95 completed independent research studies and published reports, many of which were randomly controlled trials to ensure rigorous testing and clinically validated results. That’s why over 200 Department of Defense (DOD) practitioners and over 92 Veterans Administration (VA) hospitals use Alpha-Stim with military personnel to treat acute, chronic, and post-traumatic pain, anxiety, depression, and insomnia.
Proven to Significantly Reduce Pain, Anxiety, Depression, and Insomnia.
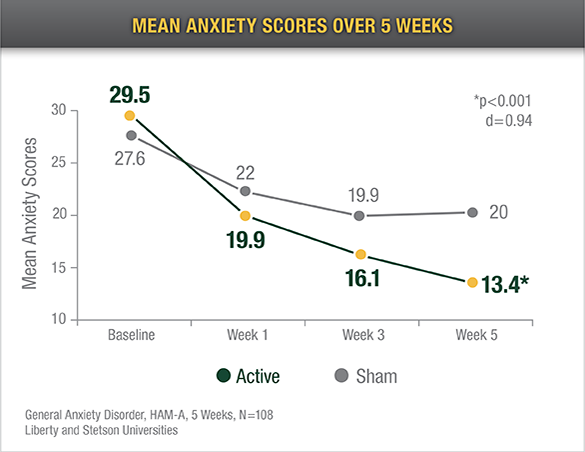
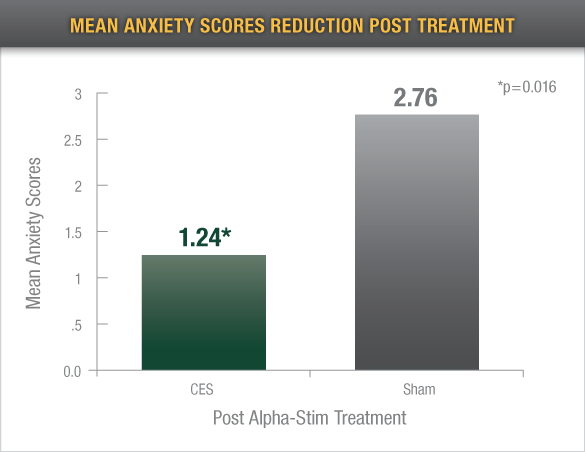
Reduced Anxiety
Eighty-three percent (83%) of the active Alpha-Stim CES group reported a decrease in anxiety of ≥ 50% on the HAM-A from baseline to endpoint of study
In a study of preoperative anxiety, Alpha-Stim recipients reported significantly less anxiety after one 20 minute application5
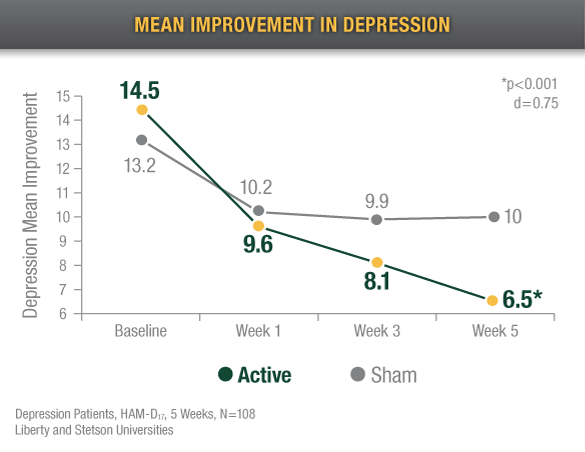
Reduced Depression
Alpha-Stim CES recipients reported an 82% decrease in depression after 5 weeks of treatment4
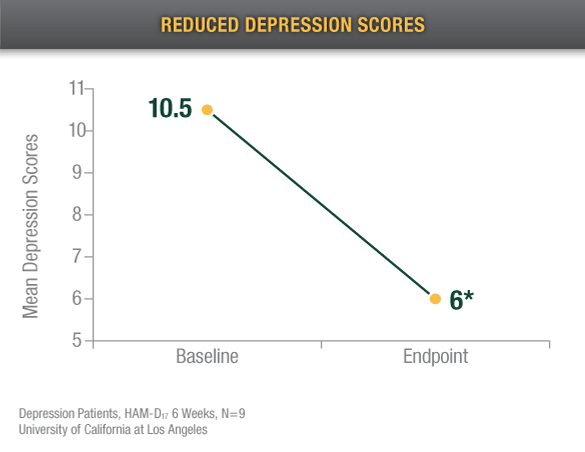
After 6 weeks of treatment, Alpha-Stim users experienced 42.8% improvement
Safety
Alpha-Stim has proven its safety in 95 research studies, published reports and ongoing research, physician and patient surveys. Scores of professionals who have used Alpha-Stim technology have remarked on the unsurpassed safety and quality of the devices.
In over 30 years of studies involving 8,800 people, only minor side effects have been reported. The two most common side effects were headaches (0.10%) and skin irritation at the electrode sites (0.07%, only seen in light-skinned people).
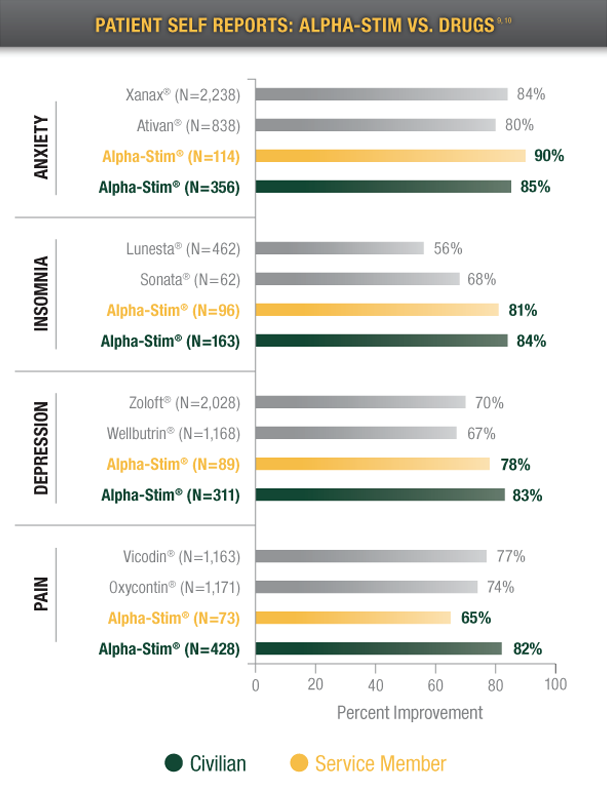
Patients who reported a positive response, according to: 2011 WebMD Drug Surveys, 2011 Alpha-Stim Service Member Survey (N=152), and 2011 Alpha-Stim Civilian Patient Survey (N=1,745).9,10
More information:
I’m Here To Help!
Press the button below to get in touch with me.